The home pregnancy test is a bit of a marvel. Cheap and quick, it provides vital information to women in the privacy of their homes. New diagnostic health tests that are similarly inexpensive and easy to use could be a boon for public health, and some chemists have turned to a surprisingly flimsy material for help: paper.
Paper-based sensors could make it easier for people around the world to assess their own health, helping with the in-home management of chronic conditions such as cardiovascular disease and perhaps even facilitating the early detection of cancer and other illnesses. In countries vulnerable to dangerous contagions such as Ebola, the sensors might also provide early warnings of an outbreak.
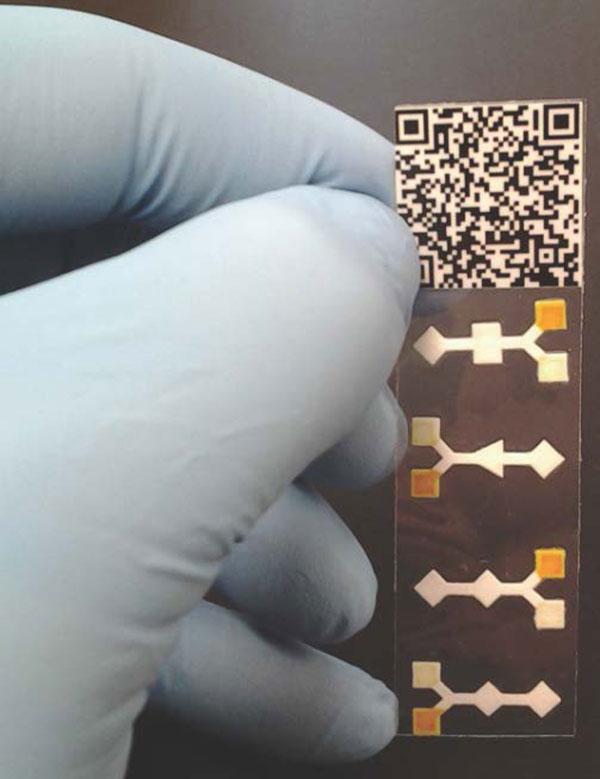
A paper-based analytic device (PAD) developed in Z. Hugh Fan’s lab at the University of Florida can detect glucose and a protein in its four test units. Samples deposited in the center of each well react with chemicals to change from yellow to brown (for glucose) or blue (for the protein). Diamond-shaped areas at each end of the units don't contain any reagents and help reveal false positives.
Paper-based tests already are used to detect problems in urine, to help diagnose influenza and strep throat, and to manage diabetes, among other applications. The continued success of these tools, which are designed to give quick results on the spot, will depend on a handful of factors, according to the World Health Organization. They’ll have to be affordable, sensitive and specific. And they’ll need to be user-friendly, fast and able to be interpreted without any other equipment. Microfluidic paper-based analytical devices, also known as micro-PADs or just PADs, fit the bill.
Essentially a tiny, self-contained lab on a handheld paper card or strip, the micro-PAD was conceived in the mid-twentieth century; more modern experimental devices were introduced in 2007. Since then micro-PAD researchers have experimented with increasingly complex designs that process samples, control the flow of fluid, amplify signals and perform other tasks. They can contain liquid samples within hydrophobic boundaries printed with wax and other materials, use three-dimensional structures to control the flow rate of samples, direct flow paths with valves that operate without instrumented controllers or actuators, deliver reagents in a predetermined order, and display test results.
Despite being made of paper, the 3D structures can be quite intricate in design. Some are inspired by the centuries-old art of origami. “They transform a simple 2D, lateral-flow test strip into a sophisticated, 3D device” for portable chemistry, says University of Florida researcher Z. Hugh Fan. Still, major technological obstacles must be overcome before micro-PADs are more widely used, including improving their sensitivity and ability to detect several biomarkers at once, Fan says.
Displaying clear test results has also proved difficult for micro-PAD designers, due partly to the innate characteristics of paper, which is opaque and autofluorescent. “Most micro-PADs are based on colorimetric detection,” Fan says. Some progress has been made by pairing the devices with smartphones to more precisely detect color changes and better interpret results.
“I am hopeful that μPADs will be used for screening infectious diseases in hospitals and in resource-poor regions in the near future,” he says.




