In the 1986 horror classic The Fly, a scientist played by Jeff Goldblum manages, quite unintentionally, to mix his biology with that of a housefly — with gruesome results.
But the real-world mutant fruit flies that scientists used to understand body patterning are almost as bizarre: Flies with legs on their brows instead of antennae. Flies with extra chest sections, complete with duplicate wings. Flies missing big chunks of their heads.
These freaky flies have something in common: They’re mixing up their head-to-tail body plans. And they earned three scientists the Nobel Prize in Physiology or Medicine in 1995.
Two of the scientists, Eric Wieschaus and Christiane Nüsslein-Volhard, conducted a now-famous genetic screen of fruit fly embryos in 1979 and 1980 while working at the European Molecular Biology Laboratory in Heidelberg, Germany. By feeding parent flies a powerful mutagen, they created a horde of larvae with genetic mistakes, including ones that affected how the fly embryo arranges bits of tissue, from head to tail, in sections — a process called segmentation. (The pair tell the tale of this landmark experiment in the 2016 Annual Review of Cell and Developmental Biology.)
The other Nobel laureate, Edward Lewis of Caltech, discovered key players, later named Hox genes, that tell these fruit fly segments and other body parts what tissues and structures they should become.
Fruit flies, it turns out, have their own segmentation path, different from ours: They make a big chunk of tissue and then slice it up, like one would a loaf of bread. In contrast, vertebrates (including humans) churn out segments one by one, like a string of sausages, as they build the tissue. But many of the genes involved — Hox and others found later — are the same.
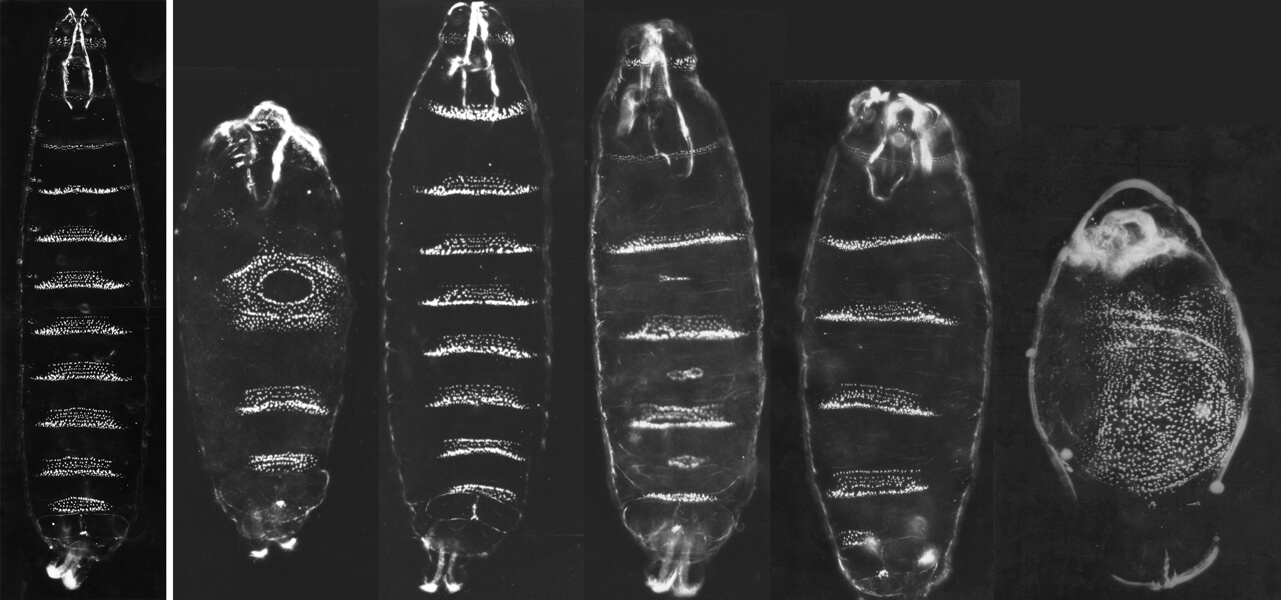
A landmark genetics screen by two scientists unearthed mutants with segmentation defects in the fruit fly Drosophila. On the left is the outer layer, or cuticle, of a normal early larva. To the right are ones of various mutants, with clear abnormalities.
CREDIT: E. WIESCHAUS & C. NÜSSLEIN-VOLHARD / AR CELL AND DEVELOPMENTAL BIOLOGY 2016
These commonalities extend to the need for a sort of “ruler” that guides segmentation and Hox actions by helping cells identify their position in the body. That ruler takes the form of a two-way gradient. Cells closest to the head end make lots of a chemical called retinoic acid, and those at the tail end make two other compounds, called FGF and Wnt. These diffuse along the body, such that different spots contain different amounts of the chemicals. So, for example, a cell that’s closer to the head than the tail will “know” its position because it’s bathed in plenty of retinoic acid, but not so much Wnt or FGF.
Pearls on a string
Vertebrate segments arise from tissue called the mesoderm. Sandwiched between the cells that will make skin and those that will make most internal organs, the mesoderm will yield tissues such as bone and muscle.
As the embryo grows, part of the mesoderm tissue near the head begins to make its segments in the form of beads of tissue called somites, one on each side of the future spinal cord. They are squeezed out of that mesoderm like toothpaste from a tube, says Robb Krumlauf, a developmental biologist at the Stowers Institute for Medical Research in Kansas City, Missouri. These will turn into vertebrae and skeletal muscles. (Other body parts will develop from cells outside of the segments.)
If the segmentation process goes wrong, vertebrae can take the wrong shape: half-vertebrae, fused vertebrae or wedge-shaped ones, for example. In people, this causes a type of scoliosis, and also may affect the kidneys, heart and other body parts.
How does the embryo make just the right number of segments, all the right size? In the 1970s, English researchers came up with a model they called “clock and wavefront.” The embryo’s clock would “tick” to indicate each time a segment should be produced. The wavefront would consist of a maturation process traveling from head to tail, and cells at the crest of that maturation wave would be ready to segment. Whenever the clock ticked, they would spit out a new segment.
The developing mammalian embryo produces two somites, one each side of the future spinal canal, every time an internal clock “ticks.” The process is guided by a protein called FGF that is made by the tail end of the embryo and diffuses along its length, forming a gradient. Somite production occurs at a spot (the wave front) where the concentration of FGF is at just the right level when the clock makes a tick. The process repeats itself over and over, gradually building up segments, from which vertebrae and skeletal muscle are made. Two other molecules, Wnt and retinoic acid, also form gradients, and with FGF these are key to telling tissues where they are along an embryo’s length.
At that time, scientists had no idea what molecules would control either clock or wavefront, or if the theory was even correct. The first hard evidence for a clock came from experiments with chicken eggs, published in 1997.
Developmental biologist Olivier Pourquié, now at Harvard Medical School, was studying the chick version of a gene called hairy that is involved in segmentation in fruit flies. He and his colleagues saw the hairy gene turn on in a cyclical manner: starting out at the tail, and then closer to the head, every 90 minutes. And every 90 minutes, the embryo made a new segment.
That study confirmed that a ticking clock did underlie segmentation, says Michalis Averof, a comparative developmental biologist at CNRS in Lyon, France. In 2012, he reported a similar oscillator in beetles.
Scientists still don’t know what sets that clock’s pace, but they now know that a variety of other proteins, including two of those ruler proteins, Wnt and FGF (and another called Notch), turn on genes like hairy. The other part of the system — the wavefront of maturation — is characterized by concentrations of FGF. Since FGF is made at the tail end, levels of the protein will be highest there and lowest at the head. Cells that have a low enough level of FGF when the clock ticks will form a segment.
Changing the speed of the clock can have profound effects on the body plan, as Pourquié found in a 2008 study on snakes. Snakes have hundreds of vertebrae, compared to the few dozen in other vertebrates like chickens, mice and humans. How did this come to be? Compared with that of a mouse, “their clock is accelerated,” Pourquié found. The faster it ticks, the more segments get made, creating the snake’s long spine. He doesn’t yet know why the snake clock ticks faster, though.
The Hox code
The bone-and-muscle segments, and the rest of the embryo’s developing tissues, need instructions so that the ones near the front make shoulders and arms, the ones at the back end make hips and legs, and so on. This process, too, depends on the ruler laid down by retinoic acid, Wnt and FGF. The position of cells with respect to the ruler tells them which Hox genes to activate. The Hox genes then turn on other genes, to make the right size and shape of vertebrae, or a tail, arm, liver, etc.
It’s complicated: Mammals have 39 different Hox genes, activated in different combinations along the body and with different parts to play. For example, mice usually grow a defined series of vertebrae, including 13 thoracic segments with ribs and six lumbar segments without. But when scientists bred mice to lack the Hox10 gene, the creatures grew little ribs on the lumbar segments. In rare cases in people, mutations in Hox genes cause diverse effects such as club foot, hair loss and extra fingers and toes.
Lewis, who worked with Hox mutant flies in the 1970s, also discovered a remarkable pattern to the Hox genes. In DNA, they are lined up in the same order in which they are produced, from head to tail, in the embryo. Genes at one end of the line spring into action in response to retinoic acid, with that signal emanating from the head; the other end responds to Wnt and FGF, signals from the rear.
A collection of genes called HOX are activated in different parts of an animal’s body plan, telling cells and tissues what to become. In the DNA, the genes line up in the same order as they are used in a developing embryo. There are remarkable similarities between the HOX genes of disparate creatures, such as fruit flies, mice and humans. In mammals, the HOX genes diversified so that there are four sets (HOX A, B, C and D) to the fly’s single set. Duplications also led to an expanded number of HOX genes in each set.
Much remains unknown about how bodies are arranged — how the same set of Hox genes creates such different body plans in different animals, for example, and how the pace of the segmentation clock sets just right to make a spine to fit a snake or a mouse or a person. Studying such things in people, of course, is difficult. So Pourquié and colleagues recently turned to human stem cells in a dish.
Using genetic trickery, they engineered the cells to flash yellow every time a certain clock gene turned on. Watching for the yellow glow, the researchers detected a clock that had five hours between each tick. Pourquié now aims to figure out just what controls that five-hour timing.
It’s astounding, Krumlauf says, how similar the parts of the body-plan system are across such a wide variety of organisms. Each animal uses many of the same genetic tools, in different ways, to create its own unique shape.
In that respect, then, it’s not so surprising that Jeff Goldblum’s character melded so completely with a fly. Wnt, FGF, Hox genes … it’s how we apply them that makes us the creatures we are.




